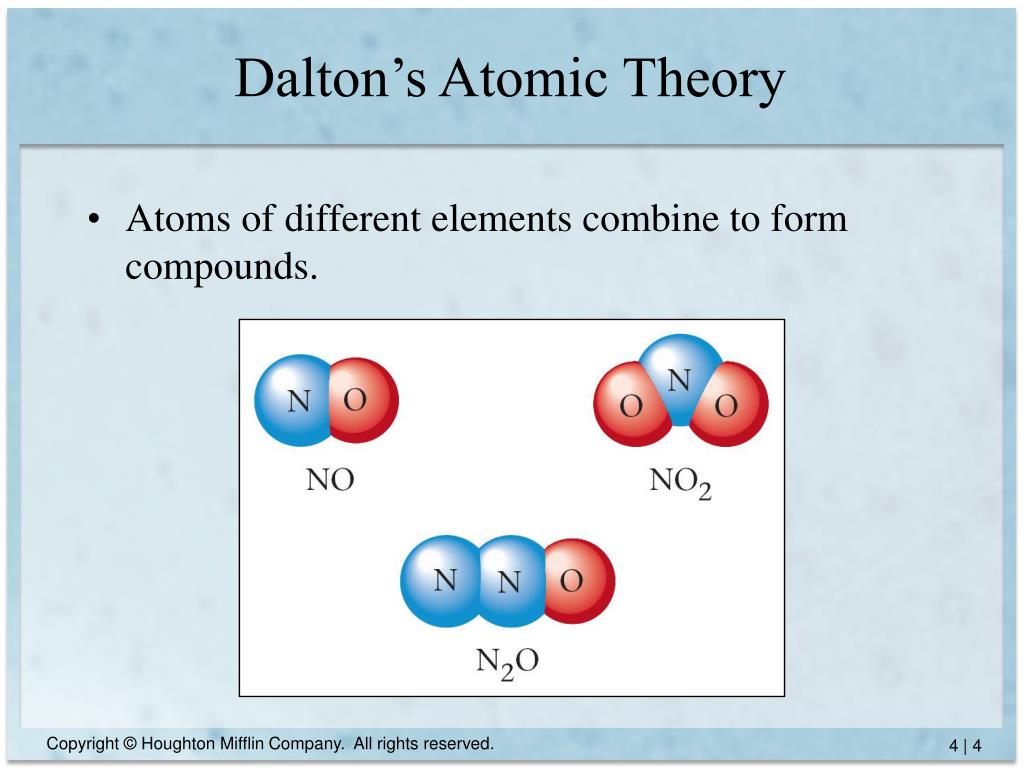
There were an infinite number of atoms, but different types of atoms had different sizes and shapes. The atomists of the time (Democritus being one of the leading atomists) believed there were two realities that made up the physical world: atoms and void. The ancient atomic theory was proposed in the 5th century bce by the Greek philosophers Leucippus and Democritus and was revived in the 1st century bce by the Roman philosopher and poet Lucretius. The word " atom" comes from the Greek \(\alpha \tau \omicron \mu \omicron \sigma\) and means "indivisible". Daltons atomic theory is the basis of the current atomic theory, though the atoms are no more considered ‘indivisible. He was a British physicist, chemist, and meteorologist who is well known for many of his contributions to the pioneering research of atoms, the law of partial pressures, Daltonism, etc. In this study guide, you can revise how the periodic table arranges elements according to their. Atoms are made from protons, neutrons and electrons. Law of conservation of mass The law of conservation of mass states that the net change in mass of the reactants and products before and after a chemical reaction is zero. Atoms of different elements are different and have different masses. National 5 Atomic structure Atomic theory. All atoms of the same element are alike and have the same mass. They cannot be divided into smaller particles, created, or destroyed. Atoms are the smallest particles of matter. (Credit: Antoine Coypel Source: (opens in new window) License: Public Domain) The introduction to the early theory of the atom was done by a scientist named John Dalton (1766-1844). Dalton’s atomic theory consists of three basic ideas: All substances are made of atoms. Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen).\): Democritus. The Law of Definite Proportions applies when elements are reacted together to form the same product. Late nineteenth- and early twentieth-century atomic models. As you read through the list above, youll see that every idea can be tested by experiment. Many consider 2008 the 200th anniversary of atomic theory, John Dalton’s momentous theory of the nature of matter.
Instead, its value lies in the research ideas it contains.

His book A New System of Chemical Philosophy (Part I, 1808 Part II, 1810) was the first application of atomic theory to chemistry. Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. Daltons atomic theory is important not because everything he said was correct. Atom - Dalton, Bohr, Rutherford: English chemist and physicist John Dalton extended Proust’s work and converted the atomic philosophy of the Greeks into a scientific theory between 18. Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. \): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B.

